GRK 2762 - Research Projects

Project T3Modulation of stroma-immune interaction to prevent RT-induced cardiac injury
Principle Investigator
Prof. Dr. med. Tienush Rassaf
Clinic for Cardiology and Vascular Medicine
University Hospital Essen
Summary
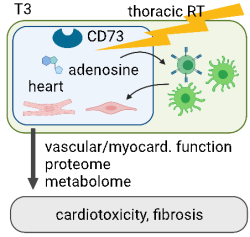
A majority of patients with advanced cancers will receive radiotherapy (RT), but the dynamic effects on the cardiovascular system remain incompletely understood. Thoracic RT for the treatment of e.g. breast and lung cancer or lymphoma comes along with an increased risk of heart failure (particularly with preserved ejection fraction), coronary artery and valvular heart disease. It should be noted that some patients appear more susceptible to RT-associated cardiac injury. The underlying reasons (e.g. predisposition, combination therapy) remain to be studied. The detrimental sequelae upon heart exposure to radiation include vascular injury followed by oedema, and dynamic activation of the inflammasome finally leading to fibrosis in the working myocardium and vasculature. A key contributor of RT-induced lung injury is an enhanced activity of CD73 leading to increased/profibrotic adenosine levels to increase activity in stromal cells. Whether this is relevant for RT-related heart injury has not been studied systematically. The applicant has recently developed a novel three-dimensional (3D), simultaneous quantitative approach to visualize heart injury components by combining bleaching-augmented solvent-based non-toxic clearing (BALANCE) with light sheet fluorescence microscopy (LSFM).1-3 Taking advantage of this approach, this projects aims to identifiy the underlying pathological CD73-dependent signaling in the heart following RT.
Selected Publications
Michel L, Mincu RI, Mahabadi, et al. Troponins and brain natriuretic peptides for the prediction of cardiotoxicity in cancer patients: a meta-analysis. Eur J Heart Fail. 2019 doi:10.1002/ejhf.1631
Totzeck M, Hendgen-Cotta UB, Lüdike P, et al. Nitrite Regulates Hypoxic Vasodilation via Myoglobin–Dependent Nitric Oxide Generation. Circulation 2012;126:325-34. doi: 10.1161/CIRCULATIONAHA.111.087155
Luedike P, Hendgen-Cotta UB, Sobierajski J, et al. Cardioprotection Through S-Nitros(yl)ation of Macrophage Migration Inhibitory Factor. Circulation 2012;125:1880-9. doi: 10.1161/ CIRCULATIONAHA.111.069104
Rassaf T, Flögel U, Drexhage C, Hendgen-Cotta U, Kelm M, Schrader J. Nitrite reductase function of deoxymyoglobin: oxygen sensor and regulator of cardiac energetics and function. Circ Res. 2007;100:1749-54. doi: 10.1161/CIRCRESAHA.107.152488
Rassaf T, Bryan NS, Maloney RE, et al. NO adducts in mammalian red blood cells: too much or too little? Nat Med. 2003;9(5):481-2. doi: 10.1038/nm0503-481

